





Akkuzid
Application instruction:
 Akkuzid – the combined drug with hypotensive and diuretic action.
Akkuzid – the combined drug with hypotensive and diuretic action.
Form of release and structure
Akkuzid is issued in the form of tablets, film coated: biconvex, pink, on both parties of risk (on 10 pieces in blisters, on 3 blisters in a cardboard pack):
- 10 mg +12,5 mg: oval, on one party marking of "PD 222";
- 20 mg +12,5 mg: triangular shape, on one party marking of "PD 220";
- 20 mg +25 mg: a round form, on one party marking of "PD 223".
Active agents as a part of 1 tablet of 10 mg +12,5 mg:
- Hinapril – 10 mg (a hydrochloride of a hinapril – 10,832 mg);
- Hydrochlorothiazide – 12,5 mg.
Active agents as a part of 1 tablet of 20 mg +12,5 mg:
- Hinapril – 20 mg (a hydrochloride of a hinapril – 21,664 mg);
- Hydrochlorothiazide – 12,5 mg.
Active agents as a part of 1 tablet of 20 mg +25 mg:
- Hinapril – 20 mg (a hydrochloride of a hinapril – 21,664 mg);
- Hydrochlorothiazide – 25 mg.
Auxiliary components (10 mg +12,5 mg / 20 mg of +12,5 mg / 20 mg of +25 mg): monohydrate of lactose – 32,348/77,196/64,696 mg; magnesium carbonate – 35,32/70,64/7064 mg; K25 povidone – 4/8/8 mg; кросповидон – 4/8/8 mg; magnesium stearate – 1/2/2 mg.
Structure of a film cover (10 mg +12,5 mg / 20 mg of +12,5 mg / 20 mg of +25 mg): Opadry pink OY-S-6937 (a hypro rod, a gipromelloza, titanium dioxide, a macrogoal 400, dye yellow and red iron oxide) – 3/6/6 mg; grass wax – 0,05/0,1/0,1 mg.
Indications to use
Akkuzid appoint for treatment of arterial hypertension at patients to whom the combined use of a hinapril and hydrochlorothiazide is shown.
Contraindications
Absolute:
- Addison's disease;
- Quincke's disease in the anamnesis as a result of the previous use of inhibitors of an angiotensin-converting enzyme (APF), a hereditary and idiopathic Quincke's disease;
- The diabetes mellitus which is followed by a chronic disease of kidneys with a renal failure of moderate severity (at a glomerular filtration rate <60 ml/min.);
- Renal (at clearance of creatinine <30 ml/min.) and a liver failure in heavy degree;
- Anury;
- Obstruction of the taking-out path of a left ventricle of heart;
- Refractory hypopotassemia, hyponatremia and hypercalcemia;
- The combined use with antagonists of receptors of angiotensin II or other inhibitors of an angiotensin-converting enzyme at a diabetes mellitus and damages of target organs in an end-stage, functional disturbances of kidneys from moderate severity to heavy degree (at a glomerular filtration rate <60 ml/min. / 1,73 sq.m), hyperpotassemias (> 5 mmol/l), chronic heart failure with the lowered arterial pressure;
- The combined use with aliskireny for patients with a diabetes mellitus, a hyperpotassemia (> 5 mmol/l), functional disturbances of kidneys (at a glomerular filtration rate <60 ml/min. / 1,73 sq.m), chronic heart failure with the lowered arterial pressure;
- Deficit of lactase, lactose intolerance, glyukozo-galaktozny malabsorption;
- Age up to 18 years (the profile of safety of Akkuzid for this age group of patients is not investigated);
- Hypersensitivity to drug components, and also to sulfonamide derivatives.
Relative (Akkuzid apply with care) in the presence of the following diseases / states:
- Therapy by diuretics in the past and observance by patients of a diet with limited consumption of salt or carrying out a hemodialysis;
- The states which are followed by decrease in volume of the circulating blood (including vomiting and diarrhea);
- The chronic heart failure in heavy degree proceeding with or without the accompanying renal failure;
- Aortal stenosis, cerebrovascular diseases (sharp lowering of arterial pressure against the background of therapy by inhibitors of an angiotensin-converting enzyme can lead to deterioration in a course of these diseases);
- Oppression of a marrowy hemopoiesis;
- Serious autoimmune system illnesses of connecting fabric (including a system lupus erythematosus, a scleroderma);
- States after transplantation of kidneys, a bilateral stenosis of renal arteries or a stenosis of an artery of the only kidney;
- Diabetes mellitus;
- Functional disturbances or the progressing liver diseases;
- Extensive surgical interventions and general anesthesia;
- Gout;
- Closed-angle glaucoma;
- Symptomatic hyperuricemia;
- Disturbances of water and electrolytic balance (hypercalcemia, hypopotassemia, hyperpotassemia, hyponatremia);
- The combined use with the cardiac glycosides and other drugs capable to lead to development of ventricular tachycardia in the pirouette type;
- The combined use with other anti-hypertensive means, and also inhibitors of mammalian Target of Rapamycin (mTOR) enzymes and dipeptidilpeptidaza (DPP)-4;
- Advanced age.
Akkuzid's use is contraindicated at pregnant women, and also when planning pregnancy. Women of reproductive age during therapy need to use reliable contraceptive methods. In the period of a lactation breastfeeding needs to be interrupted.
Route of administration and dosage
Akkuzid accept inside. Does not accord meal on efficiency of therapy of influence.
Frequency rate of reception – once a day.
The initial dose the patient who is not receiving diuretic (irrespective of, monotherapy hinaprily was carried out or not), makes 10 mg +12,5 mg. In need of further the dose can be increased to maximum – 20 mg +25 mg. Effective control of arterial pressure is, as a rule, reached at Akkuzid's use in the range of daily dose from 10 mg +12,5 mg up to 20 mg +12,5 mg.
The initial dose at functional disturbances of kidneys of easy severity (at clearance of creatinine> 60 ml/min.) makes 10 mg +12,5 mg.
To appoint Akkuzid as initial therapy to patients with renal failures with clearance of creatinine <60 ml/min. does not follow. At functional disturbances of kidneys of moderate severity (clearance of creatinine – 60-30 ml/min.) hinaprit appoint in an initial dose 5 mg with further titration.
Correction of the mode of dosing is not required to elderly patients.
Side effects
In general, the patients receiving therapy hinaprily in combination with a hydrochlorothiazide had passing the disturbances which were found and poorly expressed. They do not depend on race, a sex, age and duration of therapy.
At more than 1% of patients such side reactions were observed: sleeplessness, rhinitis, dizziness, a headache, unproductive persistent cough (after the termination of therapy passes independently), cough, a mialgiya, increased fatigue, viral infections, nausea, vomiting, upper respiratory tract infections, bronchitis, dyspepsia, an adynamy, vazodilatation symptoms, pharyngitis, thorax pains, вертиго, an abdominal pain and to back.
In 3% and 4% of cases at the patients receiving the combined therapy increase in concentration of creatinine and an urea nitrogen was observed (respectively).
The side effects developing at the same time with Akkuzid's use which are observed in 0,5-1% of cases:
- Cardiovascular system: heart failure, a heart consciousness, tachycardia, the expressed lowering of arterial pressure, orthostatic hypotension, disturbances of a cordial rhythm, a myocardial infarction, an ischemic stroke, peripheral hypostases (including generalized), stenocardia, a faint, hypertensive crisis;
- Alimentary system: dryness of a mucous membrane of an oral cavity and throat, diarrhea or lock, pancreatitis, meteorism, gastrointestinal bleeding, disturbance of functional hepatic tests, hepatitis;
- Skeletal and muscular and connecting fabric: joint pains;
- Hemopoietic system: thrombocytopenia, hemolitic anemia, leukopenia, agranulocytosis;
- Reproductive system: decrease in a potentiality;
- Nervous system: hyperexcitability, paresthesia, drowsiness, depression;
- Urinary system: renal failures, infections of uric ways, acute renal failure;
- Respiratory system: sinusitis, short wind;
- Organ of sight: vision disorder;
- Allergic reactions: photosensitization, skin rash, itch, Quincke's disease, small tortoiseshell, anaphylactic reactions, Quincke's disease of intestines, mnogoformny exudative erythema, pemphigus, exfoliative dermatitis, Stephens-Johnson's syndrome;
- Other reactions: a hyperpotassemia, an alopecia, the increased sweating.
The disturbances developing at use of a hydrochlorothiazide:
- Cardiovascular system: orthostatic hypotension, arrhythmias, vasculitis;
- Alimentary system: sialadenitis, cholecystitis, anorexia;
- Hemopoietic system: aplastic anemia;
- Allergic reactions: a necrotic vasculitis, a purpura, photosensitivity, a respiratory distress syndrome (including not cardiogenic hypostasis of a lung and a pneumonitis), anaphylactic reactions up to shock;
- Reproductive system: decrease in a potentiality;
- Urinary system: intersticial nephrite, disturbance of renal function;
- Organ of sight: passing acute myopia, passing illegibility of sight, acute open angle glaucoma;
- Metabolism: hypomagnesiemia, hypopotassemia, hypercalcemia; the gipokhloremichesky alkalosis (in the form of dryness of a mucous membrane of an oral cavity, thirst, disturbances of a heart rhythm, changes in mood or mentality, spasms and muscle pains, nausea, vomiting, increased fatigue or weakness) can cause a hepatic coma or hepatic encephalopathy; a hyponatremia (in the form of confusion of consciousness, spasms, a lethargy, delay of process of thinking, increased fatigue, excitability, muscular spasms), a glucosuria, a hyperuricemia with development of an attack of gout, a hyperglycemia. Therapy of a tiazidama can lower tolerance to glucose, and the diabetes mellitus latentno proceeding can demonstrate. Use of high doses can lead to increase in concentration of lipids in a blood plasma.
The disturbances recorded as a result of carrying out post-market researches:
- Alimentary system: hepatitis, digestive tract tumors, cholestatic jaundice, vomiting, esophagitis, diarrhea;
- Cardiovascular system: deep vein thrombosis, bradycardia, pulmonary heart, vasculitis;
- Skeletal and muscular system: miositis, myopathy, arthritis;
- Respiratory system: bronchial asthma, pneumonia;
- Urinary system: albuminuria, hamaturia, pyuria, nephrosis;
- System of a hemopoiesis: anemia;
- Nervous system: meningism phenomena, gait disturbance, hemiplegia, paralysis, disturbances of the speech, amnesia;
- Integuments: small tortoiseshell, petechias, makulo-papular rash;
- Metabolism: weight reduction;
- Other reactions: generalized hypostases, shock, inflammation of a hypodermic fatty tissue, accidental injuries, hernia.
Special instructions
- Chronic heart failure: perhaps excessive lowering of arterial pressure, proceeding with an oliguria, an azotemia and, in rare instances, an acute renal failure and even a lethal outcome (therapy needs to be begun under careful control of the specialist);
- Quincke's disease: in cases of development of guttural whistle or a Quincke's disease of a glottis, language or the person, difficulties at breath or a proglatyvaniye of food it is necessary to cancel immediately therapy and to appoint adequate treatment. For decrease in symptoms it is possible to use antihistaminic drugs. Medical observation is required before disappearance of hypostasis. At damages of a throat the lethal outcome is possible. At the paraglossa, a glottis or a throat menacing with development of obstruction of respiratory tracts it is necessary to carry out the adequate emergency treatment including hypodermic administration of solution of Epinephrinum (adrenaline) 1:1000 (0,3-0,5 ml). Development of a Quincke's disease of intestines is also possible (it is diagnosed by means of a computer tomography of belly area, ultrasonography or at the time of performing surgery). After the treatment termination symptoms usually disappear. At the combined use with inhibitors of mTOR enzymes and DPP-4 the probability of development of a Quincke's disease is higher;
- Arterial hypotension: development of symptomatic hypotension is the most probable at the reduced volume of the circulating blood. Passing arterial hypotension a contraindication to further reception of Akkuzid is not, however correction of his dose can be required;
- Agranulocytosis: development of an agranulocytosis and suppression of a marrowy hemopoiesis arises at patients with uncomplicated arterial hypertension at renal failures, in particular with diseases of connecting fabric more often (control of number of leukocytes in blood is necessary). In cases of emergence of any symptoms of an infection it is necessary to consult immediately at the doctor as it can be manifestation of a neutropenia;
- System lupus erythematosus: its aggravation is possible;
- Functional disturbances of kidneys: it is not necessary to appoint Akkuzid at clearance of creatinine less than 30 ml/min. as therapy can promote progressing of an azotemia. At clearance of creatinine of 30-60 ml/min. drug is appointed in lower initial dose;
- Influence of RAAS: suppression of activity of RAAS in certain cases can lead to functional disturbances of kidneys, at heavy heart failure development of an oliguria and/or the progressing azotemia, and in some cases – to an acute renal failure and/or a lethal outcome is possible;
- Double blockade of activity system renin-angiotensin-aldosteronovoy (SRAA): manifestation signs – a lowering of arterial pressure, a hyperpotassemia and changes in function of kidneys (including an acute renal failure). It is necessary to avoid simultaneous use RAAS-aktivnykh of means with Akkuzid;
- Stenosis of renal arteries: perhaps reversible increase in level of an urea nitrogen of blood and serumal creatinine;
- Abnormal liver function: development of a hepatic coma is possible;
- Water and electrolytic balance: to reveal possible disturbances of electrolytic balance it is necessary to control serumal levels of electrolytes regularly. Development of a hyperpotassemia is possible (risk factors – renal failures, a diabetes mellitus and the combined use with the kaliysberegayushchy diuretics, drugs of potassium and/or substitutes of salt containing potassium). The combined use of kaliysberegayushchy diuretics with Akkuzid is not recommended. Against the background of a hypopotassemia the toxic effect of cardiac glycosides can also amplify (risk factors – cirrhosis, an artificial diuresis, inadequate consumption of the drugs improving metabolism of a myocardium, the accompanying therapy by glucocorticosteroids or adrenocorticotropic hormone, the combined use with the drugs increasing risk of development of a hypopotassemia against the background of reception of thiazide diuretics). At patients with peripheral hypostases the hyponatremia can develop in hot weather;
- Diabetes mellitus: disturbance of control of concentration of glucose in a blood plasma is possible. Decrease in content of potassium in a blood plasma can lead to increase in tolerance to glucose;
- Cough: usually it is resistant, unproductive, after the end of therapy passes independently;
- Surgical interventions: development of arterial hypotension is possible, is eliminated with increase in volume of the circulating blood;
- The volume of the circulating blood: at insufficient consumption of liquid development of an extraordinary lowering of arterial pressure is possible;
- Closed-angle glaucoma and acute myopia: the main symptoms arising within the first hours-weeks after the beginning of reception of Akkuzid – eye pain or sharp decrease in sight. Closed-angle glaucoma without the corresponding therapy can lead to irreversible loss of sight. It is necessary to cancel immediately Akkuzid and, if necessary, to carry out the emergency medical or surgical intervention.
During treatment, especially at the beginning, at control of vehicles or performance of other works requiring special attention it is necessary to be careful.
Medicinal interaction
At the combined use of Akkuzid with some drugs / substances the following effects can develop:
- Lithium drugs: decrease in renal clearance and increase in serumal concentration of lithium, development of symptoms of lithium intoxication and increase in risk of development of undesirable effects (the combination is not recommended, in need of the combined use it is necessary to be careful);
- The tetracycline and other medicines interacting with magnesium: decrease in their absorption;
- Ethanol, narcotic analgetics or barbiturates: strengthening of probability of orthostatic hypotension;
- Not depolarizing muscle relaxants: strengthening of their action;
- The drugs increasing the content of potassium in blood: development of a hyperpotassemia is possible;
- Diuretics: strengthening of anti-hypertensive action;
- Hypoglycemic medicines: increase in tolerance to glucose;
- Non-steroidal anti-inflammatory drugs: weakening of a diuretic, anti-hypertensive and natriuretic effect;
- Glucocorticosteroids, adrenocorticotropic hormone: strengthening of loss of electrolytes, in particular potassium;
- Other anti-hypertensive means: strengthening of their action, in particular ganglioblokator or beta adrenoblockers;
- Pressor amines: insignificant reduction of action of adrenomimetik;
- Warfarin (at single use): change of anticoagulating effect;
- Digoxin: increase in risk of development of symptoms of intoxication by digoxin;
- Medicines for treatment of gout: disturbance of control of a condition of patients with gout;
- Aliskiren, antagonists of receptors of angiotensin II or other inhibitors of an angiotensin-converting enzyme (at the diseases/states specified in contraindications): the combination is not recommended;
- Ion-exchange resins: decrease in absorption of a hydrochlorothiazide;
- Allopyrinolum, cytostatic means, immunodepressants, procaineamide: increase in risk of development of a leukopenia;
- Drugs with hypotensive action, narcotic analgetics, medicines for the general anesthesia: strengthening of anti-hypertensive action of a hinapril;
- The cardiac glycosides and other drugs capable to lead to development of ventricular tachycardia in the pirouette type: increase in probability of development of a hypopotassemia and strengthening of toxic effect of these drugs (the combination demands care);
- Gold drugs (intravenous administration of an aurotiomalat of sodium): development of the symptom complex including a hyperemia of the person, vomiting, nausea, a lowering of arterial pressure and a grippopodobny syndrome;
- Inhibitors of DPP-4 and mTOR enzymes: increase in risk of development of a Quincke's disease (the combination demands care).
Terms and storage conditions
To store in the place, unavailable to children, at a temperature up to 25 °C.
Period of validity – 3 years.
Name of drug
Price
Drugstore
Akkuzid тбл п / about 20 mg +25mg No. 30, Goedecke GmbH
523 rub.
 Network of the Moscow drugstores of IFC
Network of the Moscow drugstores of IFCAkkuzid тбл п / about 20 mg +12.5mg No. 30, Goedecke GmbH
563 rub.
 Network of the Moscow drugstores of IFC
Network of the Moscow drugstores of IFCAt regular visit of a sunbed the chance to develop a carcinoma cutaneum increases by 60%.

Mushrooms - the surprising inhabitants of our planet having a set of wonderful qualities. Thanks to one of them, a mold mushroom of Penici...
Section: Articles about health
Use of medicinal plants in therapy is urgent today, more than ever. The drugs made of curative herbs cannot replace completely modern synthetic drugs, but their use becomes frequent serious help in simplification a leak...
Section: Articles about health
The number of long-livers is very small. One person from 5 thousand lives up to age of 90 years, and the centenary boundary steps over only one of 20 thousand. However, doctors claim that each of us is quite able to affect own destiny. At the same time it is not so much about living as long as possible, how many about an opportunity to keep physical and intellectual activity and to avoid decrepitude. We will also talk about the ways helping to achieve this result today....
Section: Articles about health
Helminthosis is one of the most widespread diseases. Statistically, with any species of helminths it is infected porridges...
Section: Articles about health
Bees – really unique beings. Practically all products of their life activity are used by the person. Since the most ancient times medicinal properties of honey and other substances received in the course of beekeeping are known. The fact that all these пр is especially significant...
Section: Articles about health
The majority of gynecologic diseases prove three main signs, each of which speaks about need of a visit to the gynecologist. Certainly, it is possible to establish the exact diagnosis only after inspection, but on the basis of some signs it is possible to assume existence of this or that pathology. Let's consider symptoms of the female diseases which are found most often....
Section: Articles about health
Contrary to popular belief, the multiple sclerosis (MS) is not connected neither with sclerous changes of walls of vessels, nor about age...
Section: Articles about health
Impossibility to conceive the child – a trouble of many Russian families. During quite long time was considered that main "culprits" of troubles such are women. Modern physicians claim that the situation is different: about a half of failures at...
Section: Articles about health
Practice of hypnotic impact on consciousness of the person contains about two millennia. During this time scientists managed to learn a lot of things about a phenomenon of hypnosis and learned to facilitate a condition of the patients having heavy illnesses with its help....
Section: Articles about health
Residents of big cities quite often have a disease which is known as the syndrome of chronic fatigue (SCF) today. This illness...
Section: Articles about health
Urogenital candidiasis (milkwoman) – a fungal infection which annoys unpleasant feelings in the field of generative organs, being followed by white curdled allocations, an itch, discomfort during an urination, pain. She is called by Candida fungus – a mustache...
Section: Articles about health
The list of stereotypes of which, apparently, all know strongly includes following: British surely eat porridge for breakfast. Perhaps, not all modern residents of Britain arrive quite so, but for those from them which continue to follow this tradition, it is possible to be glad sincerely: oat flakes are a product which regular use not only helps the person to keep force and beauty long. Porridge in a special way influences an organism, protecting it from seriousness...
Section: Articles about health
On the head of the person about one million hair follicles, or as they are called still, hair bulbs are located. At the moment he is born...
Section: Articles about health
Doctors claim that the people not so familiar with a dorsodynia occur among adult Russians very seldom. At the same time the vast majority of the patients who are periodically testing this indisposition do not hurry to ask for medical care at all. With one St...
Section: Articles about health
The climax, or menopause is the normal process of the termination of genital function of the woman which is followed by serious hormonal changes in an organism. Usually the menopause begins at the age of 50-55 years, but characteristics of this process are very individual. Factors of earlier approach of a climax are irregular sex life, numerous abortions, addictions, existence of endocrine, autoimmune and gynecologic diseases, frequent stresses and excessive hobby of diets...
Section: Articles about health
The unpleasant feelings connected with spring breakdown are familiar almost to each of us. Often happens that in March-April on the person...
Section: Articles about health
Small appetite at the child – the complaint which pediatricians should hear practically from each mother. Most often it is carried to the category of children's whims, however the refusal of food in certain cases can be to alarming symptoms therefore it cannot be ignored....
Section: Articles about health
Scientists always aimed to offer fundamental explanations for medical problems. Their theories formed the basis of modern methods of treatment of the hardest pathologies and helped to save a set of lives. However stories are known also such theoretical constructions, following to which brought to mankind of a trouble and torture, ruined destinies and health of many people....
Section: Articles about health
History of cultivation of a buckwheat contains more than five thousand years. Grain which is received from this plant is used for пригото...
Section: Articles about health
To look healthy and means well-groomed not only to be pleasant to people around, but also to feel strong, sure and taken place. Specialists in the field of cosmetology quite often note that whether not all women are able to look after skin...
Section: Articles about health
Life activity of one-celled fungi of the sort Candida, related to yeast is a proximate cause of development of candidiasis (milkwoman). Normal these microorganisms are a part of the microflora living in an oral cavity and intestines of most of people and also in a female genital tract. The pathological phenomena are observed when fungi begin to breed too violently. At the same time there is an inflammatory process affecting mucous membranes and which is shown very nepr...
Section: Articles about health
Zone hypostases under eyes - very widespread problem giving to people is a lot of inconvenience. Hypodermic fabric in these parts having...
Section: Articles about health
All are familiar with cold, and practically everyone believes that he has sufficient knowledge and experience that correctly to treat it. In practice most of people makes mistakes in attempts to get rid of rhinitis, and divides numerous delusions it....
Section: Articles about health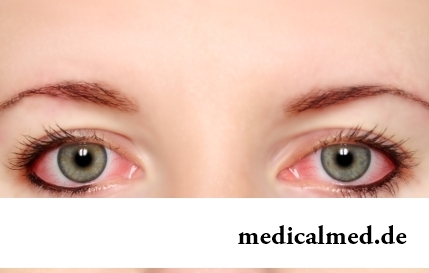
The sclera and mucous membrane of an eye are intensively supplied with blood vessels which problem - to saturate nervous tissues of body with nutrients and oxygen. In a normality vessels are almost not noticeable, however at their expansion (owing to thinning of walls) become visible, painting a sclera in red color. Quite often red eyes - the signal of any trouble in an organism caused as external irritants, allergens, and diseases which need in about...
Section: Articles about health
For residents of the countries of Southeast Asia various algas are an obligatory component of a daily diet. Their priest...
Section: Articles about health
New year, wedding, birthday, office party – an occasion to drink at the Russian person will always be. How to reduce a negative impact of alcohol by an organism and to avoid a condition of strong intoxication? The most correct council – to refuse the use spirits напитк...
Section: Articles about health
About 20% of the population of our planet have a hypertension (permanent increase in arterial pressure). This disease has an adverse effect on the standard of living, reduces working capacity, and in the absence of systematic treatment threatens with such complications as a myocardial infarction, a stroke and other heavy illnesses which can result in disability or sudden death. Most of patients for maintenance of pressure at more or less acceptable level accept appointed doctors лекарст...
Section: Articles about health

