Хиберикс
Application instruction:
Structure of Hiberiks
1 dose of Hiberiks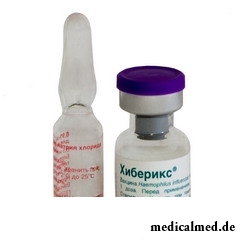 contains 10 mkg of the capsular polysaccharide emitted from a strain of Hib 20, 752 containing about 30 mkg of anatoxin. As excipients are used: lactose - 10,08 mg on 1 dose. Solvent: chloride sodium solution of 0,9% - 0,5 ml on 1 dose.
contains 10 mkg of the capsular polysaccharide emitted from a strain of Hib 20, 752 containing about 30 mkg of anatoxin. As excipients are used: lactose - 10,08 mg on 1 dose. Solvent: chloride sodium solution of 0,9% - 0,5 ml on 1 dose.
Description
Хиберикс is a vaccine against Haemophilus influenzae (V), is issued in the form of powder or the mass of white or gray-white color.
Indications to use
Хиберикс it is applied to prevention of hemophilic b of infections, children since age have 6 weeks and up to 5 years. Хиберикс has no preventive effect on other Haemophilus influenzae types and bacterial meningitis of other etiology.
Contraindications
Individual intolerance of components of a vaccine, including tetanic anatoxin. Manifestation of allergic reactions to the vaccines Haemophilus influenzae (B) entered earlier.
At acute diseases - infectious and noninfectious, and also at chronic diseases in a stage of an aggravation carrying out an inoculation not less than in a month after recovery is admissible. At the respiratory, intestinal and other infections proceeding benign carrying out an inoculation on condition of normalization of body temperature is admissible.
To patients with HIV infection use of Hiberiks is allowed.
Dosage of Hiberiks and application instruction
Age from 6 weeks to 6 months: the inoculation consists of three repetitions at an interval of 1, either 1,5, or 2 months. In various countries different schemes of vaccination are accepted. The revaccination is carried out once on the second year of life. It is recommended to combine an inoculation of Hiberiks with vaccination against whooping cough, poliomyelitis, tetanus and diphtheria.
Age from 6 months to one year: the inoculation consists of two repetitions with an interval of 1 month. The revaccination becomes once on the second year of life. Combination of vaccination by Hiberiks with vaccination against whooping cough, poliomyelitis, tetanus, a rubella, diphtheria, parotitis and measles is admissible.
Age from 1 year to 5 years: single vaccination becomes, introduction of a vaccine is made intramusculary. To patients with pathologies of coagulant system of blood it is entered subcutaneously.
Before Hiberiks's use it is necessary to examine attentively the solvent attached to it on existence of foreign particles, or outward change. At identification of physical particles or discoloration, solution cannot be used.
Before Hiberiks's use the solution which is in a set is brought in a bottle with a vaccine at the rate of 0,5 ml on 1 dose. Drug mixes up by stirring. Usually for full dissolution of lyophilisate enough less than 1 minute. After dissolution drug looks as colourless transparent liquid. If after hashing in drug not dissolved particles are found, or color does not correspond described, such drug cannot be used. After opening of a bottle drug has to be used within one working day. The remains have to be utilized.
Side reactions
After introduction 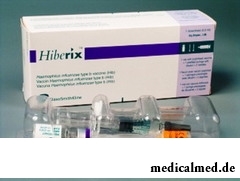 of a vaccine of Hiberiks responses from physicians show possibility in an injection site of insignificant reddenings, puffinesses and morbidities, usually no more than within 48 hours.
of a vaccine of Hiberiks responses from physicians show possibility in an injection site of insignificant reddenings, puffinesses and morbidities, usually no more than within 48 hours.
Perhaps also manifestation within 48 hours: fevers, losses of appetite, concern, vomiting, diarrhea. Similar reactions were also fixed at simultaneous vaccination by different vaccines.
According to responses of Hiberiks can cause other reactions of an organism to vaccination as:
• allergic reactions (anaphylactoid and anaphylactic), Quincke's disease;
• a hypotonic-hyporeactive episode, faints, spasms (with fever and without), drowsiness;
• apnoea;
• extremity hypostasis in an injection site, local consolidation;
• urticaria, rash.
Interaction with other drugs
Hiberiks's introduction with other vaccines is admissible. At the patients receiving immunodepressive therapy the required immune response can be not received.
Special instructions
Due to the probability of emergence in rare instances of anaphylactic reactions, after an inoculation the patient has to remain under observation of doctors not less than 30 minutes. Hiberiks's introduction intravenously is strictly forbidden!
After carrying out at premature children of vaccination by Hiberiks it is recommended to carry out by the instruction monitoring of their respiratory functions within 48-62 hours.
Storage conditions
Period of validity of substance of a vaccine of Hiberiks - 3 years. Drug with the expired term is subject to utilization. The vaccine is stored at a temperature of 2 - 8 °C in the place protected from light. Drug cannot be frozen. It is necessary to store a vaccine in the place unavailable to children.
Statistically, on Mondays the risk of getting injured of a back increases by 25%, and risk of heart attack – for 33%. Be careful.

A little more than a century ago goat milk was a traditional food stuff of most of Russians. Unfortunately, today on tables...
Section: Articles about health
The naturopathy sometimes moves as the new direction of medicine, something like fashionable hobby, and there is nothing farther from the truth. This most ancient direction, the word "naturopathy" is translated as "treatment by the nature", and, no doubt, treatment приро...
Section: Articles about health
For anybody not a secret that the modern person eats not as his ancestors. For the last 100 years in broad access there were absolutely new products which are result of use of the latest technologies in food production. Significantly ways of storage and transportation of food ingredients changed, and people of the whole world had an opportunity to regularly use those products about which their grandfathers and grandmothers did not even know....
Section: Articles about health
Producers of milk mixes for children assure: mixes are ideally balanced and adapted for needs of babies. In a sluch...
Section: Articles about health
Obesity is called a disease of 21 centuries, for the last 100 years the number of the people suffering from excess body weight considerably increased. Statistically, on Earth already about 1,5 billion corpulent people, and 500 million from them have extreme degree of completeness, are negative...
Section: Articles about health
For the city dweller the fitness is the most convenient sport. It is enough to acquire the subscription to the gym to get access to various apparatuses and an opportunity to train under the leadership of the experienced consultant. Many consider fitness the best way of maintenance of physical shape and receiving dynamic loads which the people occupied preferential with brainwork so need. Nevertheless, representations of most of consumers of similar services about специф...
Section: Articles about health
Popular joke that there are no healthy people, and is nedoobsledovanny, most of us considers an honest truth, and put that...
Section: Articles about health
Ayurveda - the most ancient tselitelsky practice which came to us from India. It represents the doctrine about maintenance of physical, psychological and moral health of the person by means of the complex of procedures including a diet, cleaning of an organism, respiratory упр...
Section: Articles about health
80% of women at least once to lives complained of discomfortable feelings to breasts, consolidations and nagrubaniye. These are mastopathy symptoms. The mastopathy is characterized by change of a ratio between ferruterous and connective tissue tissues of mammary glands. It can lead to formation of cysts (a cystous mastopathy), gland consolidation (a fibrous mastopathy), or a combination of these processes (a fibrous and cystous mastopathy)....
Section: Articles about health
All the known slogan "Protect Men!" arose not from scratch. In a sense, the nature created representatives of strong...
Section: Articles about health
It is known that the person for 80% consists of water which participates in all processes of an organism. The person loses liquid daily – as a result of sweating, breath, an urination, and its insufficient completion due to various reasons can bring to обезвожив...
Section: Articles about health
What will only not be thought up by persons interested to have a beautiful figure. Here the last innovation – for weight loss needs to be eaten greasy food. Let's understand whether there is at a fatty diet common sense....
Section: Slideshow
All know that self-treatment is dangerous. However absolutely it is almost impossible to do without it. Rate of modern life not on...
Section: Articles about health
Since the moment when the child becomes a school student, his sight begins to be exposed to the strengthened loadings which are supplemented with viewing of animated films and long computer games. During this period of life of the child development not completely created bodies to a zra...
Section: Articles about health
For residents of the countries of Southeast Asia various algas are an obligatory component of a daily diet. Their popularity is connected not only with high tastes, but also with numerous curative properties. Russians are a little familiar with products such that is distressing: algas are so useful that they should be eaten as often as possible. Let's get acquainted closer with useful properties of this seafood....
Section: Articles about health
It would seem, to buy drugs in Moscow does not make a problem – a drugstore, and not one, is available for each resident of the capital in step a toast...
Section: Articles about health
Eyes – unique body on the structure thanks to which the person obtains about 80% of information on the world around: about a form, color, size, the movement, and also many other parameters of objects or phenomena. But whether much we know about the most valuable body...
Section: Articles about health
Very often as a source of the infection which caused a disease serves our house - the place which a priori has to be safe. However disease-producing bacteria can perfectly feel not only in insanitary conditions, but also in our apartment if not to carry out due care of favourite places of their dwelling. What they − sources of their reproduction? Let's consider 10 most widespread places in our house, the most dangerous from the point of view of infection with microorganisms....
Section: Articles about health
The summer of this year in Russia was very ambiguous. Regions suffered from a merciless heat, from pouring rains, from times...
Section: Articles about health
Shops of household appliances offer us the huge choice of various devices for the house. Whether there are among this abundance devices which not only facilitate house work, but also help to keep health of the person? Of course, and we will tell about them today....
Section: Articles about health
The brain of the person is studied not one hundred years, but the quantity of the riddles connected with this body increases rather, than decreases. Perhaps, numerous delusions concerning a structure and functioning of a brain are explained by it, many of which arose long ago, but continue to exist and today. Such we are ready to acquaint readers with the most widespread myths....
Section: Articles about health
One of the useful properties presented to the person by the nature is ability to feel fear. This ability is called a signal...
Section: Articles about health
Olive oil – the product capable to make a powerful contribution to health of the person if it includes it in the diet. The rich vitamin composition of oil does it by a product number one from many diseases including from deadly. Only two tablespoons...
Section: Articles about health
The varicosity has familiarly many, statistically, this disease more than a half of all adult population. As a rule, the varicosis affects preferential superficial vessels, and is shown by characteristic cosmetic defects. The deep vein thrombosis as this illness at the initial stages can imperceptibly proceed is represented much more dangerous, and in the started cases threatens with serious danger – thrombosis. This state, when the blood clot formed...
Section: Articles about health
For the time being the perspective of heart diseases seems to most of people remote and foggy. But sooner or later практичес...
Section: Articles about health
Helminthosis is one of the most widespread diseases. Statistically, any species of helminths infected every third inhabitant of the planet. Most of specialists even consider these data strongly underestimated: some uninvited "cohabitants" of N...
Section: Articles about health
Season of activity of viral infections in the heat. Everyone can get sick, but probability of this unpleasant event it is possible and it is necessary to minimize. There is a number of rules, following to which will help or to avoid absolutely infection with flu or a SARS, or to have an illness benign and without essential complications. About ways of prevention of seasonal infections the speech in this article will also go....
Section: Articles about health