





Adelfan-Ezidreks
Application instruction:
 Adelfan-Ezidreks – the combined antihypertensive with arteriodilatiruyushchy, sympatholytic, diuretic action.
Adelfan-Ezidreks – the combined antihypertensive with arteriodilatiruyushchy, sympatholytic, diuretic action.
Form of release and structure
Dosage form – tablets: from almost white till white color, without cover, a round form, flat with slanted edges, with marking of "CIBA" on one party and dividing risky between the letters "A" and "F" – on another (on 10 pieces in the blister, in a cardboard pack of 1, 2, 3, 5 or 25 blisters).
Content of Adelfan-Ezidreksa active ingredients is 1 tablet:
- Digidralazina the sulfate hydrated –10,25 mg (on an anhydrous basis – 10 mg);
- Reserpinum – 0,1025 mg;
- Hydrochlorothiazide – 10 mg.
Auxiliary components: starch corn, attracts (Mannitolum), magnesium stearate (magnesium stearate), dinatrium эдетат, talc (magnesium hydroxysilicate).
Indications to use
Use of Adelfan-Ezidreksa is shown to patients with arterial hypertension.
Contraindications
- Age up to 18 years;
- Pulmonary heart or the isolated right ventricular insufficiency against the background of pulmonary hypertensia;
- Heart failure, the expressed tachycardia at high cordial emission (including a thyrotoxicosis);
- Heart failure because of obstruction in a left ventricle of the taking-out path (including chronic cardial compression, an aortal or mitral stenosis);
- The stratifying aortic aneurysm;
- Electroconvulsive therapy;
- Depression (including the anamnesis);
- Epilepsy;
- Parkinson's disease;
- System lupus erythematosus;
- Ulcer colitis;
- Period of an aggravation of a peptic ulcer of a stomach and duodenum;
- Heavy renal failure (the clearance of creatinine is less than 30 ml/min.), anury;
- Pheochromocytoma;
- The accompanying therapy by monoamine oxidase inhibitors (MAO) or their use in 14 days prior to purpose of drug;
- Pregnancy period;
- Hypersensitivity to related to Reserpinum to substances, gidrazinoftalazina, sulphonamide derivatives and components of drug.
Cannot appoint tablets the patients who had a myocardial infarction to a complete recovery of indicators of a hemodynamics.
With care it is recommended to appoint Adelfan-Ezidreks to patients at heart failure, coronary heart disease (or suspicion to it), a sinus bradycardia, disturbances of conductivity of heart, coronary and cerebral atherosclerosis, a peptic ulcer of a stomach and duodenum in the anamnesis, a liver failure, cholelithiasis, erosive gastritis, a diabetes mellitus, gout, bronchial asthma, disturbance of water and electrolytic balance, a porphyria, clinical manifestations of a hyperuricemia, in case of the accompanying therapy inhibitors of an angiotensin-converting enzyme (APF), blockers angiotensin II of receptors, renin inhibitors, the patient of advanced age, at planned surgery.
Route of administration and dosage
Pill is taken inside, washing down with water, daily in at one time days.
The dose is selected individually. Treatment should be begun with use of the minimum dose, depending on its therapeutic effect it is possible to raise gradually, but is not more often than once in 2-3 weeks.
The daily dose should not exceed 1-2 tablets (the maximum dose in 2 tablets is accepted in 2 receptions).
In the absence of adequate control of the arterial pressure (AP) treatment it is necessary to reconsider and appoint other drug.
Because of risk of excess accumulation of drug in an organism with care it is necessary to appoint Adelfan-Ezidreks at a renal failure and a liver, the dose and an interval between administration of drug should be established taking into account portability and ABP loudspeakers.
The special attention is required by correction of the mode of dosing at purpose of drug to elderly patients.
Side effects
As Adelfan-Ezidreks is the combined means, emergence of side effects can be caused by one of its active ingredients.
Digidralazina sulfate can become the reason of the undesirable phenomena:
- From cardiovascular system: often – heartbeat, tachycardia; sometimes – the expressed decrease in the ABP, rushes of blood to the person, stenocardia; seldom – heart failure, hypostases;
- From a liver: seldom – an abnormal liver function, jaundice, hepatitis;
- From digestive tract: sometimes – nausea, vomiting, the dispeptic phenomena, diarrhea;
- From a nervous system: often – a headache; sometimes – dizziness; seldom – anorexia, excitement, paresthesias, irritability, concern, peripheral neuritis, a condition of alarm (at purpose of a pyridoxine these side effects pass); separate cases – oppressed mood;
- Immunopathological and allergic reactions: seldom – a volchanochnopodobny syndrome;
- From system of a hemopoiesis: seldom – a leukopenia, anemia, thrombocytopenia;
- Others: sometimes – an arthralgia; seldom – fever, weakness, weight reduction of a body, a skin itch, rash.
Undesirable effects characteristic of Reserpinum:
- From cardiovascular system: sometimes – hypostases, a sinus bradycardia; seldom – pain behind a breast, arrhythmia (stenocardia symptoms), rushes of blood to the person, orthostatic hypotension; separate cases – disturbance of cerebral circulation, heart failure, a syncope;
- From digestive tract: sometimes – the increased salivation, dryness in a mouth, diarrhea, increase in secretion of a gastric juice; seldom – increase in appetite, nausea, vomiting, formation of ulcers; separate cases – gastrointestinal bleeding;
- From a respiratory organs: sometimes – an asthma, a rhinedema; separate cases – nasal bleeding;
- From urinogenital system: seldom – disturbance of an ejaculation and potentiality; separate cases – a glomerulonephritis, a dysuria;
- From sense bodys: sometimes – dacryagogue, a conjunctiva hyperemia, sight misting; separate cases – a hearing disorder;
- From a nervous system: sometimes – a depression, increased fatigue, dizziness, dreadful dreams, irritability; seldom – a headache, extrapyramidal disturbances (including parkinsonism), disturbance of concentration of attention, a condition of alarm, orientation disturbance, a stupor; separate cases – brain hypostasis;
- Metabolism and endocrine system: sometimes – increase in body weight; seldom – a gynecomastia, a galactorrhoea, increase in secretion of prolactin; separate cases – swelling of mammary glands;
- Others: seldom – an itch, eczema, decrease in a libido; separate cases – anemia, a purpura, thrombocytopenia.
Against the background of reception of a hydrochlorothiazide development of the following side effects is possible:
- From cardiovascular system: sometimes – orthostatic hypotension (which aggravation happens at simultaneous use of sedatives, anesthetics, alcohol intake); seldom – arrhythmia;
- Metabolism and water and electrolytic balance: often (generally against the background of reception of high doses) – increase in maintenance of lipids in blood, a hypopotassemia; sometimes – a hyperuricemia, a hyponatremia, a hypomagnesiemia; seldom – a glucosuria, a hypercalcemia, a hyperglycemia, at patients with a diabetes mellitus – strengthening of metabolic disturbances; separate cases – a gipokhloremichesky alkalosis;
- From the alimentary system: sometimes – nausea, vomiting, appetite loss; seldom – a lock, diarrhea, discomfort, jaundice, an intra hepatic cholestasia; separate cases – pancreatitis;
- Dermatological reactions: sometimes – skin rash, a small tortoiseshell; seldom – a photosensitization; separate cases – a toxic epidermal necrolysis, a necrotic vasculitis, manifestations on skin of a volchanochnopodobny syndrome, a lupus erythematosus aggravation;
- From system of a hemopoiesis: seldom – thrombocytopenia, sometimes with a purpura; separate cases – an agranulocytosis, hemolitic anemia, a leukopenia, oppression of a marrowy hemopoiesis;
- From sense bodys and nervous system: seldom – a vision disorder (especially in an initiation of treatment), sleep disorders, a headache, a depression, dizziness, paresthesias;
- Others: sometimes – impotence; separate cases – reactions of hypersensitivity, disturbance from respiratory system (including a pneumonitis, a fluid lungs).
Special instructions
Because of risk of suicide actions in case of symptoms of a depression administration of drug should be stopped. Provoked by Reserpinum (at high doses of Adelfan-Ezidreksa) the depression can proceed during the long period after drug withdrawal.
Presence of a digidralazin can cause a delay of water and sodium in an organism and to become the reason of hypostases, decrease in a diuresis.
Patients with abnormal liver functions need to provide systematic control of water and electrolytic balance as even its insignificant disturbances can lead to a hepatic coma, especially at cirrhosis.
The probability of development of a volchanochnopodobny syndrome is in direct dependence on a dose and the period of use of drug therefore the long maintenance therapy requires purpose of a minimal effective dose. A symptom of easy forms of a syndrome is the arthralgia which can be followed by skin rashes and fever. After drug withdrawal the condition of the patient is normalized independently. Signs of a clinical picture of a system lupus erythematosus are characteristic of hard cases.
Treatment digidralaziny should be accompanied with definition of an antinuclear factor regular (every 6 months) in a blood plasma, and in case of its detection to register credits and to watch their dynamics. In case of development of clinical manifestations of a volchanochnopodobny syndrome, immediate cancellation of Adelfan-Ezidreksa is required.
Because of existence of Reserpinum it is necessary to cancel drug before performing electroconvulsive therapy not later than in 7 days.
When carrying out planned surgery it is necessary to warn the anesthesiologist about administration of drug as preliminary cancellation of the combined means does not give a guarantee of stability of a hemodynamics during operation, perhaps expressed decrease in the ABP.
The hydrochlorothiazide causes development of a hypopotassemia, especially in patients with quickly developing diuresis, cirrhosis, deficit of intake of potassium with food, simultaneous therapy with glucocorticosteroids, stimulators of adrenocorticotropic hormone or beta adrenoceptors. The hypopotassemia can exponentiate response of heart to toxic influence of medicines of a foxglove and increase sensitivity of a myocardium. For timely definition of disturbances of electrolytic balance administration of drug should be accompanied with regular control of content of electrolytes in a blood plasma. At emergence of nausea, thirst, dryness in a mouth, weakness, concerns, drowsiness, muscular weakness, muscular pains and spasms, decrease in the ABP, tachycardia, an oliguria (nonspecific symptoms of disturbance of electrolytic balance) it is necessary to see a doctor.
Against the background of use of thiazide diuretics development of a hypercalcemia and hypophosphatemia is possible.
Use of thiazide diuretics can lead to a hypomagnesiemia as they promote the increased removal of magnesium with urine.
At high doses of a hydrochlorothiazide at the patient development of decrease in tolerance to glucose is possible that causes risk of increase in level of concentration in plasma of triglycerides, cholesterol, uric acid.
Reserpinum influences laboratory indicators in urine 17 of hydroxycorticosteroids and 17 ketosteroids a colorimetric method, underestimating their result.
Adelfan-Ezidreks exerts impact on the speed of psychomotor reactions, especially at the beginning of use. It is necessary to warn the patients managing vehicles and mechanisms about need of respect for care.
Medicinal interaction
Because of big risk of development of heavy side reactions during treatment by Adelfan-Ezidreksom the concomitant use of any medicines is recommended to be begun only after consultation of the doctor.
Terms and storage conditions
To protect from children.
To store in the place protected from moisture and light at a temperature up to 30 °C.
Period of validity – 4 years.
The 74-year-old resident of Australia James Harrison became blood donor about 1000 times. It has a rare blood group which antibodies help to survive the newborn with a severe form of anemia. Thus, the Australian saved about two million children.

For anybody not a secret that the modern person eats not as his ancestors. For the last 100 years in broad access appeared with...
Section: Articles about health
Many parents of children at the age of 2-4 years face excessively whimsical behavior of the child. The kid exhausts constant crying and whims not only the parents, but also himself. In what the reasons of children's whims. And how to fight with them?...
Section: Slideshow
During foot walks blood moves on vessels more actively and one and all bodies are supplied with a large amount of oxygen. It affects the state of health of the person very positively....
Section: Slideshow
The brain of the person is studied not one hundred years, but the quantity of the riddles connected with this body increases rather, than reducing...
Section: Articles about health
From sexual contacts each person can test insufficiently strongly expressed sexual desire or lack of satisfaction from time to time. However when it happens regularly, it is an occasion to think about health. Most of people does not hurry an obrashcha...
Section: Articles about health
All diseases from nerves – in this joke a big element of truth, are said by doctors. Constant stresses lead to decrease in protective forces of an organism, and it becomes vulnerable for a set of diseases. It is wrong to think that the stress is a problem of the present. Life of people and hundred, and one thousand years ago also abounded with problems therefore need of a relaxation understood in ancient times – to some techniques more than one thousand years. The person needs knowledge of how it is possible to relax, this knowledge пригод...
Section: Articles about health
The trophic ulcer is not an independent disease. This heavy complication arising owing to a thermal injury (a burn...
Section: Articles about health
Stroke (acute disorder of cerebral circulation) – one of the most widespread neurologic diseases. Annually in the world more than 6 million people die of this illness. From the survived patients about 80% become disabled people, and nearly a thirds from them впо...
Section: Articles about health
The next flu epidemic leads to the next panic, from year to year we give in on these manipulations: professionally alarming voice of the announcer in news, reports with calculation of the died patients, an interview with people in white dressing gowns and advertizing of anti-influenza means of different degree of inefficiency. All this reminds the Hollywood movies of epidemics threatening to destroy our planet. However, there is also one more similarity to cinema: everything comes to an end well. So, how to deal with the events, not in...
Section: Articles about health
Reactive pancreatitis - the disease which is characterized by inflammatory process in a pancreas which arises more often everything...
Section: Articles about health
Cystitis, or inflammation of a mucous membrane of a bladder, this very widespread disease which, owing to some features of a structure of bodies of urinogenital system, women have approximately four times more often than men. In the main risk group...
Section: Articles about health
From the failure of work of immune system which is shown in the form of an allergy, statistically, more than 40% of the population of the globe suffer. In most cases pathological reactions cause the substances which are contained in food stuffs, hair of animals, medicines, goods of household chemicals, cosmetics, pollen of plants, etc. On the one hand, the disease such is capable to spoil quite thoroughly to the person life....
Section: Articles about health
Modern footwear is extremely various. It stopped being only protection for legs long ago. Today shoes, boots, barefoot persons in...
Section: Articles about health
The majority of gynecologic diseases prove three main signs, each of which speaks about need of a visit to the gynecologist. Certainly, it is possible to establish the exact diagnosis only after inspection, but on the basis of some signs it is possible пр...
Section: Articles about health
The immunity role in growth of the child is invaluable. The proteins-immunoglobulins produced by immune system preserve the child against the diseases capable − owing to an organism weak still − to serve as a stressful factor, to become the reason of many complications and delays in development of the kid. If the immune system weakened, health of the child is under direct threat and needs active actions for strengthening of protective forces of an organism − preferably non-drug....
Section: Articles about health
Tick-borne encephalitis – one of the most dangerous viral diseases which causative agents transfer and is given to people by ixodic mites. Эт...
Section: Articles about health
Epilepsy is one of widespread neurologic diseases. Parents, whose children suffer from this illness, should face rumors and delusions, many of which remained since the Middle Ages....
Section: Articles about health
The phenomenon of the panic attack is known long ago, but the reasons of its emergence still are up to the end not found out. It is established that more than 30% of people at least once in life become the victims of very unpleasant phenomenon: without everyones on that the reasons they have a feeling of horror which is followed by a cardiopalmus, a shiver and the fever or feeling of sudden heat increased by sweating, breath constraint, dizziness, nausea....
Section: Articles about health
The drugs stopping or oppressing life activity of pathogenic microorganisms are widely applied in clinical practice with 4...
Section: Articles about health
Life expectancy in various regions of Earth is not identical. Social stability, economic wellbeing, availability and level of medical care, household comfort, literacy of the population in the field of observance sanitary гигиен exert impact on it...
Section: Articles about health
Dark circles (bruises) under eyes – a shortcoming with most of which often fight against the help of cosmetics (proofreaders, saloon procedures and so forth), eliminating only its visibility. However, according to doctors, skin around eyes – the indicator of many disturbances in an organism. To reveal them at early stages, without having disguised bruise, and having addressed its reasons – a task of each person who is regularly finding under with own eyes dark stains. Early detection and elimination of the disease lying in wasps...
Section: Articles about health
Each person has easy indispositions which he transfers "standing", trying not to ask for medical care. Argu...
Section: Articles about health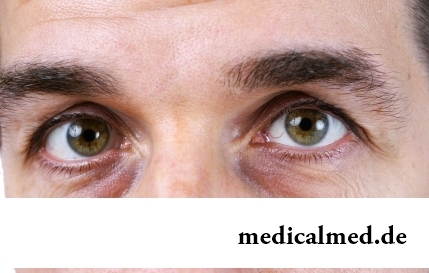
Zone hypostases under eyes - very widespread problem giving to people is a lot of inconvenience. Hypodermic fabric in these parts has very loose structure and almost does not contain collagenic fibers. Besides, the skin covering подглазья constantly is exposed...
Section: Articles about health
Long time antibiotics were considered as a panacea from all diseases and were appointed even at insignificant symptoms of an infection. Even now not everyone knows in what force of antibiotics how and when they should be accepted. Let's discredit 7 popular myths about such drugs....
Section: Articles about health
History of mankind contains several tens of epidemics whose emergence was compared by eyewitnesses and historians to doomsday. With...
Section: Articles about health
New year, wedding, birthday, office party – an occasion to drink at the Russian person will always be. How to reduce a negative impact of alcohol by an organism and to avoid a condition of strong intoxication? The most correct council – to refuse the use spirits напитк...
Section: Articles about health
The chia plant, or the Spanish sage, is from South America. The indigenous people of the continent since ancient times used its seeds in food: small, but very nutritious kernels, in a form the reminding fasolina. Indians knew about useful properties of seeds of a chia, and applied them to maintenance of vitality and increase in endurance before serious exercise stresses....
Section: Articles about health
