





Valtsit
Application instruction:
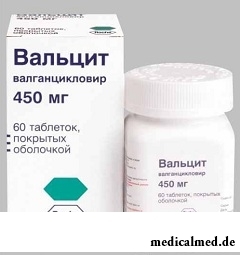 Valtsit – the drug relating to group of antiviral means.
Valtsit – the drug relating to group of antiviral means.
Form of release and structure
Is issued in the form of tablets, film coated (in bottles polyethylene on 60 pieces, 1 bottle in a cardboard pack).
Active ingredient – a valgantsiklovira a hydrochloride, in 1 tablet of 450 mg.
Auxiliary components: cellulose microcrystallic – 47,8 mg; кросповидон – 23,9 mg; K30 povidone – 23,9 mg; stearic acid – 6 mg.
Fillers of a film cover: titanium dioxide (E171), ferrous oxide red (E172), a gidroksipropilmetiltsellyuloz, polysorbate, polyethyleneglycol, water cleared.
Indications to use
Valtsit it is appointed at treatment of a Cytomegaloviral retinitis at patients about AIDS, and also for the purpose of prevention of a Cytomegaloviral infection after organ transplantation.
Contraindications
- Number of thrombocytes less than 25 000 C / мкл;
- Clearance of creatinine less than 10 ml/min.;
- Absolute number of neutrophils less than 500 C / мкл;
- Concentration of hemoglobin is lower than 80 g/l;
- Hypersensitivity of the patient to a gantsiklovir, a valgantsiklovir, and also to any of drug components (reactions of cross sensitivity to Valtsit, a valatsiklovir and an acyclovir are possible that is explained by a similar chemical structure of these drugs);
- Period of pregnancy and lactation;
- Age less than 12 years.
With care drug is appointed to patients with a renal failure, and also to elderly people (efficiency and safety for this age category are not established).
Route of administration and dosage
Drug is recommended to be accepted inside during food.
In order to avoid overdose it is necessary to adhere to the dosing mode recommended by the instruction strictly:
- Induction therapy to patients with an active Cytomegaloviral retinitis – within 3 weeks to accept 900 mg 2 times a day. At long induction therapy the risk of a miyelotoksichnost increases;
- Maintenance therapy after performing induction therapy to patients with an inactive Cytomegaloviral retinitis – 900 mg of 1 times a day. In case of deterioration in a current of a retinitis repeated carrying out a course of induction therapy is allowed;
- Prevention of a Cytomegaloviral infection after organ transplantation – to accept from 10 to 100 days after transplantation, 1 time a day 900 mg.
In the course of treatment of the patients having a renal failure it is necessary to control carefully the level of creatinine of serum or the clearance of creatinine (CC). For men the clearance of creatinine is calculated depending on its level in serum on the following formula: (140 – age (years)) × body weight (kg)) / 72 × 0,011 × creatinine of serum, µmol/l. For women of KK = 0,85 × an indicator for men. Further calculation for men is given:
- At KK of 10-24 ml/min. the dose for induction therapy makes 450 mg each 2 days, for a maintenance therapy – 450 mg 2 times a week;
- At KK of 25-39 ml/min. a dose for induction therapy – 450 mg of 1 times a day, for a maintenance therapy – 450 mg each 2 days;
- At KK of 40-59 ml/min. a dose for induction therapy – 450 mg 2 times a day, for a maintenance therapy – 450 mg of 1 times a day;
- At KK from 60 ml/min. a dose for induction therapy – 900 mg 2 times a day, for a maintenance therapy – 900 mg of 1 times a day.
It is not necessary to appoint Valtsit to the patients who are on a hemodialysis (KK less than 10 ml/min.).
At a neutropenia, thrombocytopenia, oppression of marrow, a heavy leukopenia, anemia, a pancytopenia, aplastic anemia it is not recommended to begin therapy if concentration of thrombocytes less than 25 000 C / мкл or the maintenance of neutrophils less than 500 C / мкл and also if the level of hemoglobin is lower than 80 g/l.
Side effects
Data of clinical trials:
At patients is after organ transplantation by the most frequent side effects arising against the background of Valtsit's reception are: tremor, hypostases of the lower extremities, dorsodynias, headaches, sleeplessness, graft rejection, nausea, vomiting, diarrhea, hypertensia. The most part of the listed manifestations carries moderately or poorly expressed character.
Among the most often found side reactions at administration of drug by patients about AIDS and a Cytomegaloviral retinitis are noted: neutropenia, fever, weakness and headaches, oral cavity candidiasis, diarrhea.
Among patients after transplantation with a frequency of 2% the undesirable phenomena which were not observed among patients with a Cytomegaloviral retinitis were noted: abnormal liver function, hypertensia, hyperpotassemia, giperkreatinininemiya.
The side effects recorded more than at 5% of patients after organ transplantation and with a Cytomegaloviral retinitis:
- From an organism in general: weakness, peripheral hypostases, hypostases of the lower extremities, fever, anorexia, loss of appetite, dehydration, cachexia, weight reduction, reaction of graft rejection;
- Infectious complications: oral cavity candidiasis, pharyngitis, nasopharyngitis, upper respiratory tract infection, sinusitis, pneumonia, infections of urinary tract;
- Dermatological reactions: the strengthened sweating (at night days), an acne, dermatitis, an itch;
- From an organ of sight: indistinct sight, peeling of a retina;
- From an urinary system: dysuria, renal failure, infections of uric ways;
- From the alimentary system: nausea and vomiting, lock, dyspepsia, diarrhea, ascites, abdominal pains, abnormal liver function;
- From system of a hemopoiesis: anemia, leukopenia, neutropenia, thrombocytopenia;
- From the central and peripheral nervous system: sleeplessness, tremor, paresthesias, headaches and dizziness, peripheral neuropathy, depression;
- From respiratory system: productive cough, a pleural exudate, a nasopharyngitis, pharyngitis, allocations from a nose, sinusitis, pneumonia, pneumocystic pneumonia, upper respiratory tract infections, short wind;
- From a musculoskeletal system: arthralgias, spasms in muscles, a dorsodynia, extremity pains;
- From cardiovascular system: arterial hypotension, arterial hypertension;
- Laboratory indicators: hyperpotassemia, hypopotassemia, hyperglycemia, hypocalcemia, giperkreatinininemiya, hypomagnesiemia, hypophosphatemia;
- Others: during the postoperative period complications, strengthening of a drainage of a wound, a bad wound repair, infectious complications of a postoperative wound, pain after operation can be noted.
The serious undesirable effects caused by Valtsit's reception, meeting with frequency less than 5%:
- From an urinary system: giperkreatinininemiya, decrease in clearance of creatinine;
- From the central and peripheral nervous system: mental deviations, confusion of consciousness, hallucination, agitation, spasms;
- From system of a hemopoiesis: oppression of function of marrow, pancytopenia, aplastic anemia;
- From coagulant system of blood: life-threatening bleedings;
- Others: hypersensitivity to a valgantsiklovir.
Valtsit quickly it is synthesized in ганцикловир therefore in the course of therapy the side effects characteristic of a gantsiklovir can be noted.
Special instructions
Children as its efficiency, safety and pharmacological characteristics are not established for this age category are not recommended to appoint drug.
In the course of therapy it is necessary to control regularly developed blood count with thrombocytes. To the patients suffering from a neutropenia, thrombocytopenia, a leukopenia, anemia growth factors are appointed and/or administration of drug is cancelled.
Gantsiklovir cannot be replaced with Valtsit in the ratio 1:1 as bioavailability of a gantsiklovir from Valtsit exceeds that of capsules of a gantsiklovir by 10 times. The patients transferred from capsules of a gantsiklovir have to be informed on risk of overdose if they accept Valtsin in the doses exceeding recommended.
Pilot studies on animals showed teratogenic, cancerogenic, mutagen, spermatocidal action of a gantsiklovir. Valtsit it is considered for the person potential carcinogen and teratogen which use is accompanied by risk of developing of cancer and inborn malformations. There is a probability that drug can forever or is temporary suppress a spermatogenesis.
Ability to driving of the car and control of mechanisms can be broken at emergence at the patient of such side reactions as sedations, confusion of consciousness an ataxy or dizzinesses.
The razlamyvaniye or crushing of a tablet as Valtsit – potential carcinogen and teratogen for the person is not allowed. It is impossible to allow direct contact of a tablet which integrity was broken, with mucous membranes and skin. If such contact happened, it is necessary to wash out immediately skin water with soap (eyes carefully wash out water).
Medicinal interaction
As Valtsit is synthesized in ганцикловир, the interactions characteristic of a gantsiklovir can be expected also at Valtsit's appointment.
The concomitant use of a gantsiklovir and tsilastatina/imipenem should be allowed only if the advantage of such therapy exceeds potential risk – the combined reception of these drugs can cause spasms.
AUC of a zidovudine can slightly, but is statistically significant increase (to 17%) at simultaneous oral administration of a gantsiklovir. Also the tendency (statistically doubtful) to decrease in concentration of a gantsiklovir can be noted. Some patients cannot transfer a concomitant use of both drugs in full doses as ганцикловир and a zidovudine can cause anemia and a neutropenia.
Co-administration of a mikofenolat of a mofetil and gantsiklovir can increase the maintenance of a gantsiklovir and phenolic glucuronide of mikofenolny acid.
At the patients undergoing therapy by cyclosporine after the beginning of use of a gantsiklovir some increase in the maximum level of creatinine in serum was revealed.
At at the same time reception of Valtsit and a probenetsid of patients it is recommended to inspect regarding toxicity of a gantsiklovir.
Concentration of a didanozin at simultaneous use with gantsikloviry with firmness increases therefore patients at such therapy should be observed regarding emergence of signs of toxic influence of a didanozin.
Simultaneous therapy of a gantsiklovir with other drugs breaking function of kidneys or rendering myelosuppressive effect can increase their toxic influence therefore such treatment can be appointed only in cases when the possible advantage exceeds potential risk.
Terms and storage conditions
To store at a temperature not above 30 °C in the place, unavailable to children.
Period of validity – 3 years.
Name of drug
Price
Drugstore
At regular visit of a sunbed the chance to develop a carcinoma cutaneum increases by 60%.

Bathing in broths of medical flowers and plants (phytobathtub) was eurysynusic since Cleopatra who is a good judge of everything...
Section: Articles about health
Each person knows that fervescence is an illness sign. However too low temperature (hypothermia), especially also can demonstrate existence of diseases when it is observed long enough. Such state is dangerous those...
Section: Articles about health
The healthy nutrition is the invariable principle of health and good health for long years of the woman. Nevertheless, in a diet at each stage of life there are the features allowing to support an organism by those substances which are most necessary for it at present. Eating according to them, the woman will be able to feel vigorous and strong, and also to adapt to changes in an organism so that they allowed it to lead active lifestyle at any age....
Section: Articles about health
Mushrooms - the surprising inhabitants of our planet having a set of wonderful qualities. Thanks to one of them, a mold mushroom of Penici...
Section: Articles about health
The immunity role in growth of the child is invaluable. The proteins-immunoglobulins produced by immune system preserve the child against the diseases capable − owing to an organism weak still − to serve as a stressful factor, to become the reason of many complications and delays in unless...
Section: Articles about health
Osteoporosis this general disease which main sign is decrease in density of a bone tissue. On distribution width it takes the fourth place among noninfectious diseases. The illness develops at mature age more often: in our country about a third of women and a quarter of men suffers from it 50 years are more senior....
Section: Articles about health
Radiological methods of a research are applied in medicine more than hundred years, and thanks to them millions of lives were saved. In m...
Section: Articles about health
So, you resolved to lose weight. And now you try to understand what to begin with: from exercise stresses or a diet? And how to make that process of weight loss did not give you an inconvenience, and, on the contrary, brought joy?...
Section: Slideshow
Practically each person is familiar with the annoying, pulling, unscrewing pains caused by overcooling of muscles of a back. In certain cases inflammatory process is not limited to discomfort, being followed by emergence of hypostasis, consolidations, temperature increase. At the wrong treatment the acute miositis can lead to a chronic disease or aggravation of other pathologies of a back (vertebral hernia, osteochondrosis) therefore it is important to pay attention to symptoms of an illness in time and to start to...
Section: Articles about health
The next flu epidemic leads to the next panic, from year to year we give in on these manipulations: professionally alarming goal...
Section: Articles about health
The Genetically Modified Organisms (GMO) are plants or animals (as a rule, agricultural) to whose genotype purposeful changes were made. Opposition of supporters and opponents of inclusion of such organisms in foodstuff всег...
Section: Articles about health
Several decades ago the basil (the district khan, реан, Reagan) was considered as a part of the Caucasian or east cuisine, but today it strongly took the place on tables of Russians. Greens of this plant possess a strong, pleasant smell and specific fresh taste because of which it is included almost in all dry mixes of spicy herbs, and also give to meat and fresh fish dishes....
Section: Articles about health
Eyes – one of the most vulnerable areas on a face therefore age changes concern them first of all. Whether it is possible to keep a pier...
Section: Articles about health
Feeding by a breast - the integral part of ideal motherhood allowing to come into contact with the kid and to create to it healthy immunity since early years. Nevertheless, this important process in life of mother and child can be saddened laktostazy − by a delay of milts...
Section: Articles about health
All know that self-treatment is dangerous. However absolutely it is almost impossible to do without it. Rate of modern life does not allow to handle each small trouble to the doctor and information on ways of independent delivery of health care is quite available. Means, all of us have only one: to learn to give this help competently and in those limits in which it is possible for the person who does not have vocational education....
Section: Articles about health
Statistically cystitis 25-30% of women up to 40 years have. With age this indicator raises, besides many do not get in to become...
Section: Articles about health
Statistically, at the address to doctors seven of each ten patients complain of a headache. Actually it is much more people who are periodically feeling unpleasant feelings such. Many people, apart from a headache the reason for serious fear...
Section: Articles about health
The mankind knows that some toxins at intake in the minimum quantities have therapeutic effect from an extreme antiquity. Many substances recognized poisonous are applied in the medical purposes also today, being the main operating components of the medicines which are officially produced by pharmaceutical industry. Let's tell only about the most known of them....
Section: Articles about health
Coffee - the tonic loved by many for the invigorating aroma and deep taste. Having the stimulating effect, coffee raises ра...
Section: Articles about health
Sugar - the digestible refined product which is not of special value for an organism of the modern person. The use of sugar in food is based rather on the psychological dependence caused by desire to indulge itself with something tasty, and in дальнейш...
Section: Articles about health
Practice of use of table salt in the therapeutic purposes contains not one century. Applications which do by means of the fabric impregnated with saline solution are considered especially effective. They have antibacterial and antiinflammatory effect, help to heal wounds, exempt fabrics from excess liquid. Hypertonic salt solution of potassium chloride is applied outwardly at many morbid conditions. Let's tell about the most known of them....
Section: Articles about health
Physical activity is necessary for normal functioning of a human body. At a lack of the movement cease функц...
Section: Articles about health
Many of us, probably, noticed more than once that from intellectual loadings at some point the brain as though "overheats" and "assimilation" of information is strongly slowed down. Especially this problem urgent for persons of age becomes more senior than fifty years. "It is already bad with...
Section: Articles about health
It is known that the person for 80% consists of water which participates in all processes of an organism. The person loses liquid daily – as a result of sweating, breath, an urination, and its insufficient completion due to various reasons can lead to dehydration of varying severity. Dehydration (dehydration) occurs already in case of loss of liquid in number of 1% of body weight and can result both in easy thirst, and by the death. In time to notice signs обезвож...
Section: Articles about health
Ayurveda - the most ancient tselitelsky practice which came to us from India. It represents the doctrine about maintenance physical, ps...
Section: Articles about health
For many spouses the question of planning of a family is one of the main. The problem of the choice of effective and safe contraceptives at the same time comes out on top. Russians still not often resort to operation of a vasectomy extremely popular in the USA...
Section: Articles about health
Dark circles (bruises) under eyes – a shortcoming with most of which often fight against the help of cosmetics (proofreaders, saloon procedures and so forth), eliminating only its visibility. However, according to doctors, skin around eyes – the indicator of many disturbances in an organism. To reveal them at early stages, without having disguised bruise, and having addressed its reasons – a task of each person who is regularly finding under with own eyes dark stains. Early detection and elimination of the disease lying in wasps...
Section: Articles about health

