Algerika
Application instruction:
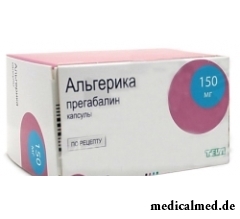 Algerika – the drug having anticonvulsant effect.
Algerika – the drug having anticonvulsant effect.
Form of release and structure
Dosage form of release of Algeriki – the capsule: opaque, on a lid the press of black TEVA color, contains in capsules partially pressed and granulated powder of white or almost white color; on 25 mg – a lid and the case of light yellow color, a text on the case – "7622"; on 50 mg – a lid and the case of light yellow color with a radial black strip, a text on the case – "7623"; on 75 mg – a lid of pink color, the case of light yellow color with a text "7624"; on 150 mg – a lid and the case of light yellow color, a text on the case – "7626": on 300 mg – a lid of pink color, on the case of light yellow color a text "7621" (on 7 pieces in blisters, on 2 or 8 blisters in a cardboard pack).
Structure of 1 capsule:
- Active agent: прегабалин – 25, 50, 75, 150 or 300 mg;
- Additional components (25/50/75/150/300 mg): talc – 9/18/8/16/32 mg, Mannitolum – 43/86/10/20/40 mg, prezhelatinizirovanny corn starch – 7/14/7/14/28 mg.
Structure of a cover of capsules:
- 25 mg "7622/TEVA": size No. 3, 48 of mg; case: titanium dioxide – 2%, dye iron oxide yellow – 0,1%, gelatin – to 100%; lid: titanium dioxide – 2%, dye iron oxide yellow – 0,1%, gelatin – to 100%;
- 50 mg "7623/TEVA": size No. 2, 61 of mg; case: titanium dioxide – 2%, dye iron oxide yellow – 0,1%, gelatin – to 100%; lid: titanium dioxide – 2%, dye iron oxide yellow – 0,1%, gelatin – to 100%;
- 75 mg "7624/TEVA": size No. 3, 48 of mg; case: titanium dioxide – 2%, dye iron oxide yellow – 0,1%, gelatin – to 100%; lid: titanium dioxide – 2,1747%, dye iron oxide red – 0,6996%, gelatin – to 100%;
- 150 mg "7626/TEVA": size No. 2, 61 of mg; case: titanium dioxide – 2%, dye iron oxide yellow – 0,1%, gelatin – to 100%; lid: titanium dioxide – 2%, dye iron oxide yellow – 0,1%, gelatin – to 100%;
- 300 mg "7621/TEVA": size No. 0, 96 of mg; case: titanium dioxide – 2%, dye iron oxide yellow – 0,1%, gelatin – to 100%; lid: titanium dioxide – 2,1747%, dye iron oxide red – 0,6996%, gelatin – to 100%.
The ink used for drawing texts on capsules: pharmaceutical glaze (shellac solution in ethanol) – 59,42%, isopropanol – 0,55%, propylene glycol – 1,3%, butanol – 9,75%, ethanol – 1,08%, dye black iron oxide – 24,65%, water ammonia – 0,001%, the purified water – 3,249%.
Indications to use
Algerika is appointed for treatment at adult following diseases:
- Fibromyalgia;
- Neyropatichesky pain;
- Generalized alarming frustration;
- The epilepsy with partial convulsive attacks proceeding with secondary generalization or without it (as additional therapy).
Contraindications
Absolute:
- Age up to 18 years;
- Pregnancy and period of a lactation;
- Hypersensitivity to drug components.
Relative (Algerik is appointed with care in the presence of the following diseases / states):
- Diabetes mellitus;
- Functional disturbances of kidneys;
- Heart failure;
- Encephalopathy (anamnestic data);
- Medicinal dependence (anamnestic data);
- The combined use with ethanol, lorazepam, oxycodone;
- Age of 65 years.
Women of reproductive age during therapy need to use effective contraceptive methods.
Route of administration and dosage
Algerika is accepted inside, regardless of meal, washing down with water in enough. Capsules need to be swallowed entirely, without crushing and without chewing.
The daily dose varies within 150-600 mg, frequency rate of reception – 2-3 times a day. Duration of treatment is defined by the doctor individually depending on indications and specific features of the patient.
The initial daily dose at all indications makes 150 mg. In 3-7 days (is defined by reaction to therapy and individual portability) its increase is possible twice. In 7 days, in case of need, its increase to maximum – 600 mg is possible.
Cancellation of treatment is carried out gradually – for at least 7 days.
At functional disturbances of kidneys at selection of a dose consider the clearance of creatinine (CC) calculated by a formula:
- Men: KK (ml/min.) = (weight in kg) × (140 - age advanced in years)/72 × concentration of creatinine in plasma (mg/dl);
- Women: KK (ml/min.) = 0,85 × KK for men.
The patient who is on a hemodialysis the daily dose of Algeriki is selected taking into account function of kidneys, at the same time directly after holding each four-hour session of a hemodialysis it is necessary to accept an additional dose (KK / an initial daily dose / the maximum daily dose / frequency rate of use):
- From 60 ml/min.: 150 mg / 600 mg / 2-3 once a day;
- 30-60 ml/min.: 75 mg / 300 mg / 2-3 once a day;
- 15-29 ml/min.: 25-50 mg / 150 mg / 1-2 once a day;
- To 15 ml/min.: 25 mg / 75 mg / once a day.
The additional daily dose appointed once after dialysis: initial – 25 mg, daily – 100 mg.
Dose adjustment for the patients having functional disturbances of a liver is not required.
To patients of 65 years because of depression of function of kidneys Algerika can be appointed in the reduced doses.
At the admission of a single dose it needs to be accepted as soon as possible. It is not necessary to accept the doubled dose.
Side effects
At Algeriki's reception it can be observed development of the following disturbances (very often (≥10%); often (≥1% and <10%); infrequently (≥0,1% and <1%); seldom (≥0,01% and <0,1%); very seldom (<0,01%, considering isolated cases)):
- Nervous system: very often – drowsiness, dizziness; often – a syncope, sedative action, irritability, decrease a libido, euphoria, confusion of consciousness, sleeplessness, a disorientation, an ataxy, a lethargy, a tremor, a memory impairment, paresthesia, amnesia; infrequently – hallucinations, an anorgazmiya, depersonalization, strengthening of sleeplessness, concern, agitation, a depression, lability of mood, difficulty in selection of words, loss of flavoring feelings, the suppressed mood, increase in a libido, dreadful dreams, the panic attacks, cognitive frustration, apathy, a hypesthesia, a nystagmus, myoclonic spasms, weakening of reflexes, a hyperesthesia, dyskinesia, burning sensation on mucous membranes and skin, psychomotor initiation, postural dizziness, a stupor, an intentsionny tremor, disturbance of the speech, coordination, balance and attention; seldom – a parosmiya, high spirits, a disinhibition, a hypokinesia, a dysgraphia;
- Alimentary system: often – a meteorism, dryness of a mucous membrane of an oral cavity, vomiting, abdominal distention, a lock; infrequently – a hypesthesia of a mucous membrane of an oral cavity, a gastroesophageal reflux, the increased salivation; seldom – a dysphagy, ascites, pancreatitis;
- Cardiovascular system: infrequently – AV blockade of the I degree, a dermahemia, inflows, increase in arterial pressure, tachycardia; seldom – sinus tachycardia, arrhythmia and bradycardia;
- Hemopoietic system: seldom – a leukopenia, a neutropenia, thrombocytopenia;
- Infectious diseases: infrequently – a nasopharyngitis;
- Urinary system: infrequently – an urine incontience, a dysuria; seldom – an oliguria, a renal failure;
- Respiratory system: infrequently – an asthma, dryness of a mucous membrane of a nasal cavity; seldom – nasal bleeding, a nose congestion, rhinitis, feeling of constraint in a drink, snore;
- Reproductive system: often – erection dysfunction; infrequently – an ejaculation delay, sexual dysfunction; seldom – a dysmenorrhea, mammary gland pains, an amenorrhea, allocations from mammary glands, and also increase them in volume;
- Skeletal and muscular system: infrequently – hypostasis of joints, twitchings of muscles, muscular spasms, constraint of muscles, a mialgiya, an arthralgia, extremity and spin pain; seldom – pain in a neck, a trachelism, рабдомиолиз;
- Skin and hypodermic fabrics: infrequently – perspiration, papular rash; seldom – a small tortoiseshell, cold sweat;
- Acoustic organ and labyrinth disturbances: often – вертиго; seldom – a hyperacusia;
- Organ of sight: often – doubling, an illegibility of visual perception; infrequently – narrowing of fields of vision, a vision disorder, decrease in visual acuity, puffiness of eyes, eye pain, quickly coming exhaustion of eyes, the raised dacryagogue, dryness in eyes; seldom – a mydriasis, irritation of eyes, flashing of "sparks" before eyes, an ostsillopsiya, squint, disturbance of perception of depth of sight, strengthening of brightness of visual perception, loss of peripheral sight;
- Metabolism and food: often – increase in weight and appetite; infrequently – a hypoglycemia, anorexia, a hyperglycemia; seldom – reduction of weight;
- Tool and datas of laboratory: infrequently – increase in activity of a kreatinfosfokinaza, aspartate aminotransferase, alaninaminotranspherase; seldom – a hypopotassemia, a giperkreatininemiya;
- Others: often – feeling of intoxication, fatigue, gait disturbance, hypostases, including peripheral; infrequently – falling, an adynamy, thirst, generalized hypostases, feeling of constraint in breasts, a fever, pain; seldom – a hyperthermia.
The side reactions recorded when carrying out post-marketing observation:
- Nervous system: with an unknown frequency – a loss of consciousness, a headache, cognitive disturbances;
- Alimentary system: seldom – nausea, a paraglossa, diarrhea;
- Cardiovascular system: with an unknown frequency – lengthening of an interval of QT, chronic heart failure;
- Respiratory system: with an unknown frequency – a fluid lungs;
- Urinary system: with an unknown frequency – an ischuria;
- Skin and hypodermic fabrics: seldom – a skin itch, a face edema;
- Organ of sight: with an unknown frequency – sight loss;
- Allergic reactions: with an unknown frequency – a Quincke's disease, hypersensitivity reaction, allergic reaction, Stephens-Johnson's syndrome.
Special instructions
During therapy or right after Algeriki's cancellation development of the epileptic status and emergence of convulsive attacks as grand mal is possible.
Disturbances from an organ of sight (in the form of decrease in visual acuity, sight loss) take place, as a rule, independently, both at treatment cancellation, and at its continuation.
There are data on development reversible (after Algeriki's cancellation) a renal failure.
After completion of treatment development of a withdrawal is possible (frequency of emergence and degree of manifestation of symptoms depend on a dose and duration of a course).
At a diabetes mellitus in case of increase in weight against the background of performing therapy there can be a need of correction of doses of hypoglycemic medicines.
In cases if symptoms of a Quincke's disease appear, drug is immediately cancelled.
During administration of drug dizziness and drowsiness can develop that increases probability of development of accidental injuries (falling) in patients of advanced age. It is necessary to be careful until the patient does not estimate possible effects of drug.
At treatment of neyropatichesky pain elderly patients have messages on developing of chronic heart failure with diseases of cardiovascular system.
The risk of development of disturbances from the central nervous system (in particular, drowsiness) increases in cases when drug is used at treatment of a pain syndrome at spine injuries (it can be connected with interaction with other medicines, including spasmolysants).
At emergence of the suicide ideas or attempts it is necessary to see a doctor immediately.
The patient with anamnestic data on medicinal dependence on any drugs Algerika is appointed with care.
Side effects in the form of encephalopathy were observed, mainly, in the presence of the accompanying states contributing to development of a disease.
In need of the combined use with opioid analgetics it is necessary to take measures of prevention of intestinal impassability and locks, in particular at elderly patients.
During Algeriki's reception it is necessary to refrain from control of motor transport and potentially dangerous works which performance demands the increased concentration of attention and bystry psychomotor reactions (it is connected with a possibility of development of side reactions in the form of dizziness, drowsiness and vision disorders).
Medicinal interaction
Pharmacokinetic interaction of Algeriki with other substances / medicines is improbable.
Pregabalin strengthens the disturbances of the main caused by oxycodone motive and mnestichesky functions, and also effects of lorazepam and ethanol.
At the combined use with opioid analgetics of Algerik can provoke weakening of function of lower parts of digestive tract, including intestinal impassability and a lock.
Terms and storage conditions
To store in the place, unavailable to children, at a temperature up to 25 °C.
Period of validity – 2 years.
There are very curious medical syndromes, for example, persuasive swallowing objects. In a stomach of one patient suffering from this mania 2500 foreign objects were revealed.
Transfusion of donor blood has almost century history. In spite of the fact that this procedure is quite usual for many people, itself п...
Section: Articles about health
The person, as well as all other beings living on our planet feels weather changing. It is the normal meteosensitivity which is not causing to healthy people of special troubles. Meteodependence, on the contrary, is morbid condition, характеризующимс...
Section: Articles about health
Healthy lifestyle today in fashion, and many parents think of that the child from the early childhood played sports. Trainings will help it to become strong and hardy, will improve coordination of movements, and also will exert positive impact on mentality: it will become more collected and purposeful....
Section: Articles about health
No, probably, the person who would not have cold. Cold, cough, a headache – these symptoms are known to everyone. Peak to Prost...
Section: Articles about health
Coffee - the tonic loved by many for the invigorating aroma and deep taste. Having the stimulating effect, coffee increases working capacity, promotes concentration of attention, fights against drowsiness and improves mood. Statistically, about 30% of inhabitants...
Section: Articles about health
Food with the increased content of sugar is attractive to most of people - it is scientifically confirmed fact. Business here not in intemperance or dissoluteness: the sweet food is associated since childhood with feeling of rest and safety which tests the kid when it absorbs maternal milk. Besides, getting into a human body, sugar strengthens production of "happiness hormones" which all of us so need. And still life of sweet teeth seldom happens cloudless: their too big loss...
Section: Articles about health
Each of us repeatedly noticed that the people having the same passport age are sometimes not similar on one-years at all. One...
Section: Articles about health
The state of health of the person depends on many factors. One of the most important is the constant, but not exhausting a physical activity. In the presence of various illnesses specialists often advise patients to do swimming which by right borrows ведущ...
Section: Articles about health
The sclera and mucous membrane of an eye are intensively supplied with blood vessels which problem - to saturate nervous tissues of body with nutrients and oxygen. In a normality vessels are almost not noticeable, however at their expansion (owing to thinning of walls) become visible, painting a sclera in red color. Quite often red eyes - the signal of any trouble in an organism caused as external irritants, allergens, and diseases which need in about...
Section: Articles about health
To look healthy and means well-groomed not only to be pleasant to people around, but also to feel strong, sure and taken place. To Spa...
Section: Articles about health
Some people consider what for medicine of the 21st century of secrets in the field of health of the person almost does not exist. It absolutely not so. The more answers scientists receive, the more the most difficult questions are raised for them by life. Besides, there are diseases, not объясн in any way...
Section: Articles about health
Frosty air, fresh wind and easy snowball at most of Russians are associated with cheerfulness, health and cheerful entertainments on which our winter is so generous. But, unfortunately, cold season sometimes brings also troubles with health. It is not about seasonal colds and frostbites, and about those chronic illnesses which symptoms are shown preferential in the winter....
Section: Articles about health
We live during an advertizing era. Daily each person receives a solid portion of persuasive councils about what to eat to be здо...
Section: Articles about health
One of the major chemical processes happening in a human body are oxidation reactions. They go with participation of fats and carbohydrates which we receive from food, and the oxygen getting to us from air. A main goal of such reactions is it is received...
Section: Articles about health
Use of medicinal plants in therapy is urgent today, more than ever. The drugs made of curative herbs cannot replace completely modern synthetic drugs, but their use becomes frequent serious help in simplification of a course of many illnesses and improvement of quality of life of chronic patients....
Section: Articles about health
Visit of doctors – business not the most pleasant, and many people do not hurry to undergo necessary planned inspections. Such behavior...
Section: Articles about health
According to doctors, more than a half of men of 25-50 years suffer from frustration of the urinogenital sphere, but the minority sees a doctor from them. And in vain - even the insignificant discomfort in the field of generative organs can serve as a symptom of an illness fraught heavy посл...
Section: Articles about health
Venereal diseases in medicine are called the infections which are transmitted preferential sexually, now they and are called - infections, sexually transmitted, or STD. Among them is also life-threatening. In spite of the fact that the majority of diseases such will respond to treatment, they are widespread everywhere, and there is no tendency to decrease in incidence. Besides, some of them promptly look younger: statistically, a third of young people at the age of 16-22 years of a str...
Section: Articles about health
Cold is such painful that each sigh becomes a victory, heat "knocks" down, and the ache in joints forces to think only about...
Section: Articles about health
When overcomes feeling of hunger, and an opportunity to have dinner fully is absent, having a snack − the meals, small on volume, stabilizing sugar level in blood comes to the rescue. The relation of nutritionists to having a snack is more often negative, but only because in кач...
Section: Articles about health
All like to sing. Small children with pleasure are engaged in a vocal, not especially thinking of hit in a melody. Adults most often hesitate, being afraid to show lack of talents in this area, and it is vain: singing is very useful for health....
Section: Articles about health
A lot of things depend on a condition of a backbone in a human body, a backbone - not only a support for a body, it also contain...
Section: Articles about health
Striya (extension) are the defects of skin having an appearance of direct or wavy strips from 1 to 10 cm long and 1-5 mm wide. In most cases at women of a striya are located on a stomach, hips, a breast or buttocks. At athletes they can appear on shoulders and внутренн...
Section: Articles about health
Hemorrhoids – extremely widespread disease. Periodically arising inflammations and bleeding of hemorrhoidal nodes cause serious discomfort to nearly fifteen percent of adults. Meanwhile, having a clear idea of the reasons of an exacerbation of an illness and following rules of precaution, it is possible to reduce substantially sharpness of unpleasant feelings and to reduce progressing of a disease....
Section: Articles about health
During foot walks blood moves on vessels more actively and one and all bodies are supplied with a large amount of oxygen. N...
Section: Slideshow
The cosmetics intended for improvement of a condition of skin, nails and hair are used by each woman. Expenses on regular acquisition of the fashionable widely advertized products of well-known companies for many become very notable and significantly to an obrema...
Section: Articles about health
Climax - process of fading of reproductive function of an organism in process of its aging. At women the main sign of its approach is the termination of a menstrual cycle. Officially the menopause is diagnosed when periods are not observed within 12 months. Age changes quite often are followed by emotional failures, disturbance of thermal control and sweating, dizzinesses and headaches, tachycardia and other unpleasant phenomena. This complex of symptoms...
Section: Articles about health